Scientists capture the first real-time 3D images of human embryo implantation, revealing dynamics that could transform infertility and IVF treatment.



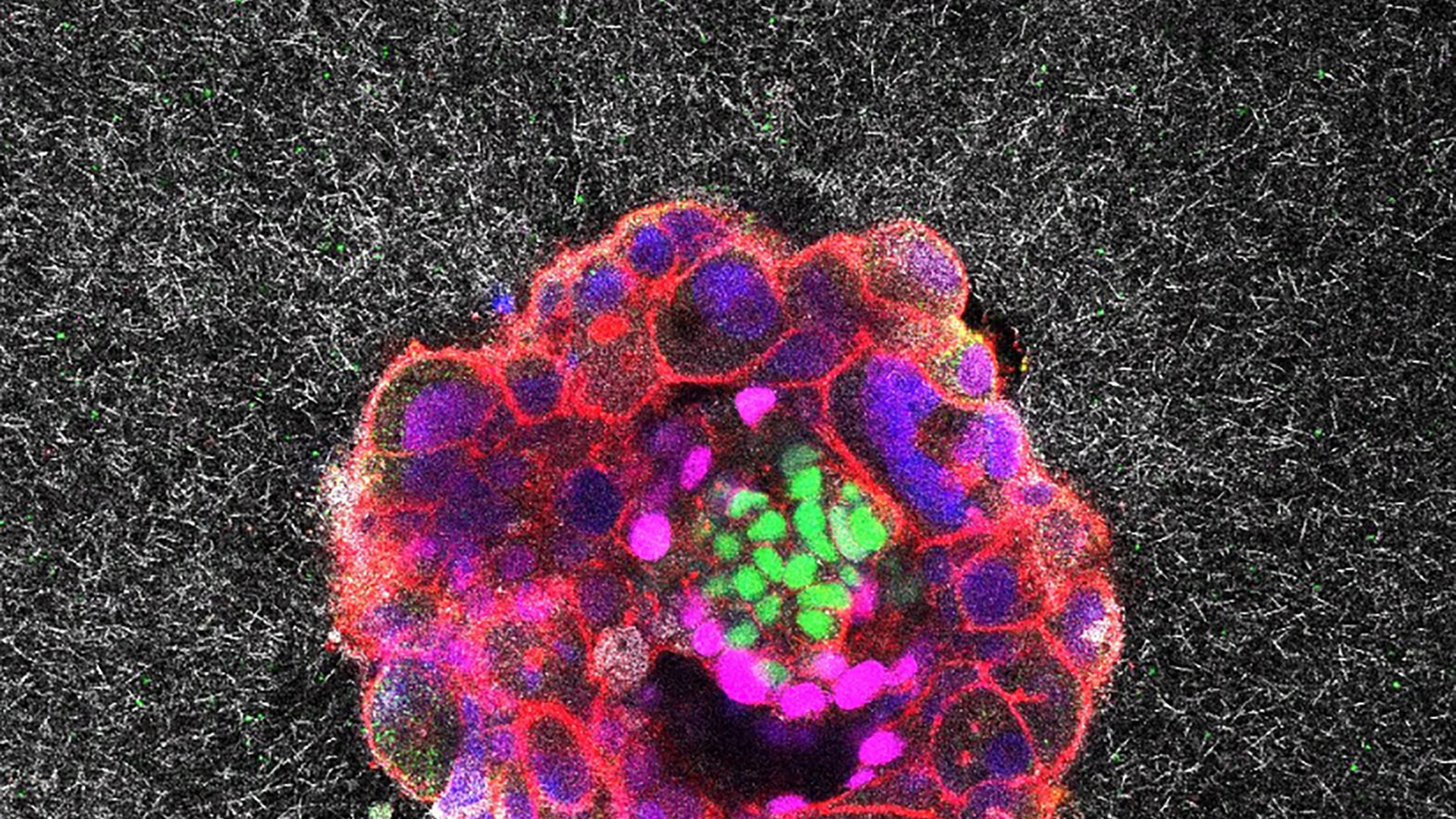
Researchers have, for the first time, captured real-time, three-dimensional images and videos of a human embryo implanting into tissue designed to mimic the human uterine environment. This groundbreaking research, largely carried out at the Institute for Bioengineering of Catalonia (IBEC) in collaboration with Dexeus University Hospital in Barcelona, has given scientists unprecedented visual access to the earliest stages of human pregnancy, offering new insight into one of medicine’s biggest mysteries: why some embryos fail to implant, causing infertility or early miscarriage.
Before this study, scientists relied on static snapshots of embryo implantation, leaving much of the process a mystery. Implantation is the pivotal event when a fertilized embryo attaches and integrates into the uterine lining, a stage responsible for about 60% of miscarriages and a leading cause of infertility. Using an innovative 3D gel platform based on collagen (a key uterine component), the IBEC team successfully visualized human embryos as they burrowed into, and radically reorganized, the uterine matrix.
Implantation failure is a dominant barrier in both natural and assisted reproduction, including in vitro fertilization (IVF). The new imaging system allows scientists and clinicians to monitor how embryos interact with a simulated uterine matrix, offering avenues to:
The researchers found that human embryos exert significant force as they burrow into the uterine tissue. This process is not passive; it’s “surprisingly invasive,” as the embryo pushes into, pulls on, and merges with its environment. The forces are vital for the embryo to invade the uterine tissue and become completely integrated, an essential step to begin a pregnancy. This intense level of activity helps explain why some women experience cramps or mild bleeding at the time of implantation.
Observing this invasive process in real time has not previously been possible, as the action takes place entirely inside the woman’s body and is shielded from direct observation. According to Samuel Ojosnegros, the lead researcher, “What happens between the transfer and the first ultrasound weeks later is a black box.” Implantation failure during this stage is responsible for approximately 60% of miscarriages and is a significant hurdle both for natural conception and in assisted reproduction like in vitro fertilization (IVF).
One of the most promising outcomes from this research lies in its potential to improve fertility treatments. Implantation remains the least understood and most failure-prone stage of both natural conception and IVF attempts. For IVF, embryos are typically transferred into the uterus five days after artificial fertilization. The events that follow, up to the first detectable pregnancy, have been largely mysterious until now.
With 3D and real-time imaging, scientists can objectively study how embryos interact with uterine tissue, both in terms of chemical signaling (like the release of enzymes that break down surrounding tissue) and biomechanics (the force exerted by the embryo). These insights can guide fertility doctors in optimizing the timing, environment, and methods for embryo transfer, potentially increasing success rates for IVF, which currently remain under 50% per cycle for most patients.
The researchers hope to use these findings to refine assisted reproduction practices and even develop treatments specifically targeting the implantation phase. By understanding what successful implantation looks like in detail, including the movements, forces, and cellular changes, clinicians can better identify what might be going wrong in cases of repeated failure and intervene more effectively.
Key technical and biological insights from species differences to mechanosensitivity underscore just how dynamic and orchestrated implantation truly is. For fertility medicine, this marks a giant step towards more personalized, evidence-based care.
