Researchers led by Hiroki Shibuya at RIKEN have uncovered how disruptions in the centrin–POC5 protein complex deform the sperm tail, offering new insight into male infertility. Using ultrastructure expansion microscopy, the team mapped sperm development in detail and showed that loss of POC5 prevents proper flagellum formation, leaving sperm nonviable.



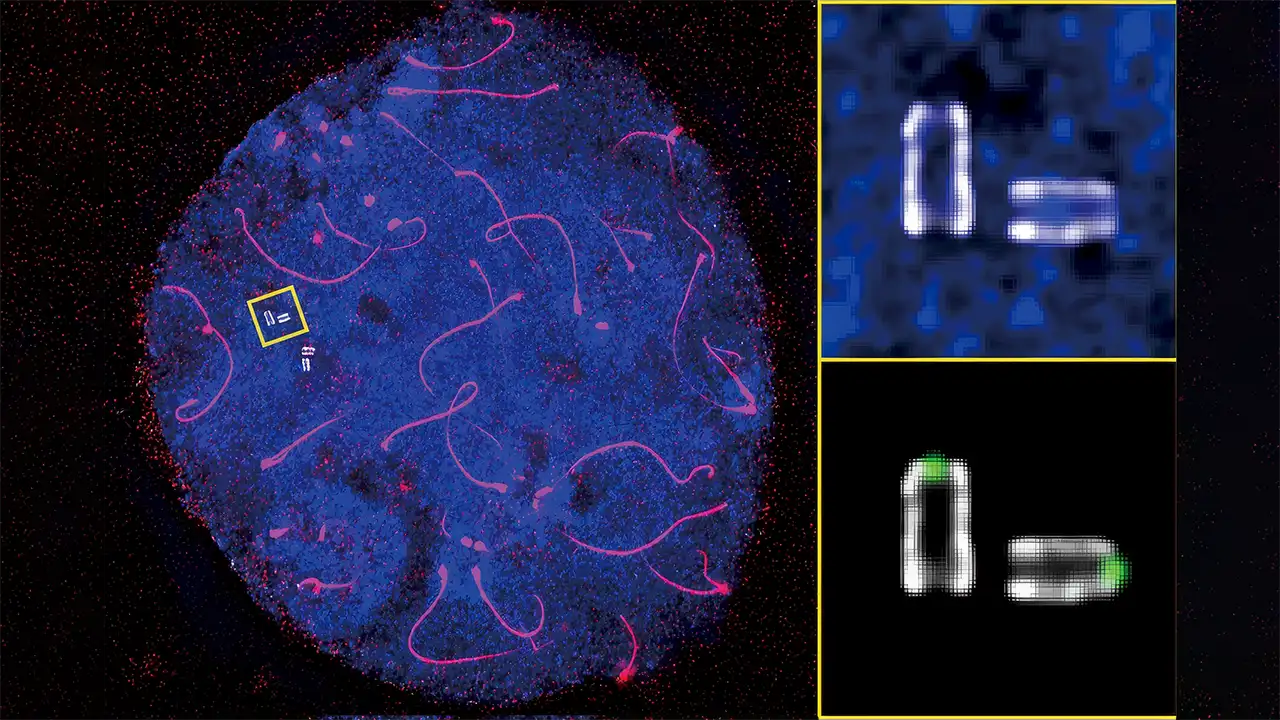
An international research team led by Hiroki Shibuya at the RIKEN Center for Biosystems Dynamics Research has identified a structural cause of sperm tail abnormalities, offering new insight into male infertility. The group used ultrastructure expansion microscopy to capture the first detailed view of the mouse flagellar base and found that disrupting a key protein complex in germ cells caused severe deformation of the sperm flagellum. Their work shows that when the distal centriole’s inner scaffold shifts, especially the centrin–POC5 protein complexes, the sperm tail cannot form as it should. The study, published in Science Advances, adds critical data to efforts to understand why male infertility remains poorly explained despite accounting for about half of all infertility cases.
The team adapted ultrastructure expansion microscopy for male mouse germ cells, first stabilizing the cells before placing them into an expandable gel. By doing so, they were able to see specific proteins and follow structural changes through spermatogenesis, something that neither electron nor fluorescent microscopy could capture on its own. Once they worked past the problems with cell movement and imaging clarity, the team was able to trace how the proximal and distal centrioles change throughout development and shape the formation of the sperm flagellum. They observed that the inner scaffold of the distal centriole strengthens following meiosis, accompanied by increased centrin–POC5 protein activity, an essential detail in preventing sperm tail abnormalities.
To understand the protein’s function, the team created a full POC5 knockout in mice using CRISPR gene editing. Although the animals appeared normal, they produced no viable sperm. Their sperm cells showed flagella that were malformed and weak, even though centriole activity in other somatic cells looked normal. The findings make clear that without POC5, sperm structure breaks down, underscoring its role in preventing sperm tail abnormalities and offering a possible explanation for some cases of human male infertility.
“While the causes of female infertility have been studied extensively,” says Shibuya, “the mechanisms underlying male infertility—which are known to account for about half of all infertility cases—remain poorly understood.” He noted that the refined imaging method may support broader clinical applications. “Our modified expansion microscopy protocol can be extended to other analyses, including human sperm, opening new possibilities for investigating fine structural abnormalities that account for male infertility,” he said. “In the long-term, this could lead to novel diagnostic and therapeutic approaches in reproductive medicine.”
