The shift in reproductive medicine toward "restorative" approaches. By targeting the molecular causes of infertility, such as oocyte aneuploidy, new biotechnologies are moving beyond embryo selection to directly improve IVF success rates and address the root biological causes of age-related fertility decline.



For over two decades, the landscape of reproductive medicine has been defined by a paradox of rising demand and plateauing biological efficacy. As socioeconomic shifts continue to delay parenthood across developed economies, the clinical community has responded with increasingly sophisticated workflows and procedural refinements.
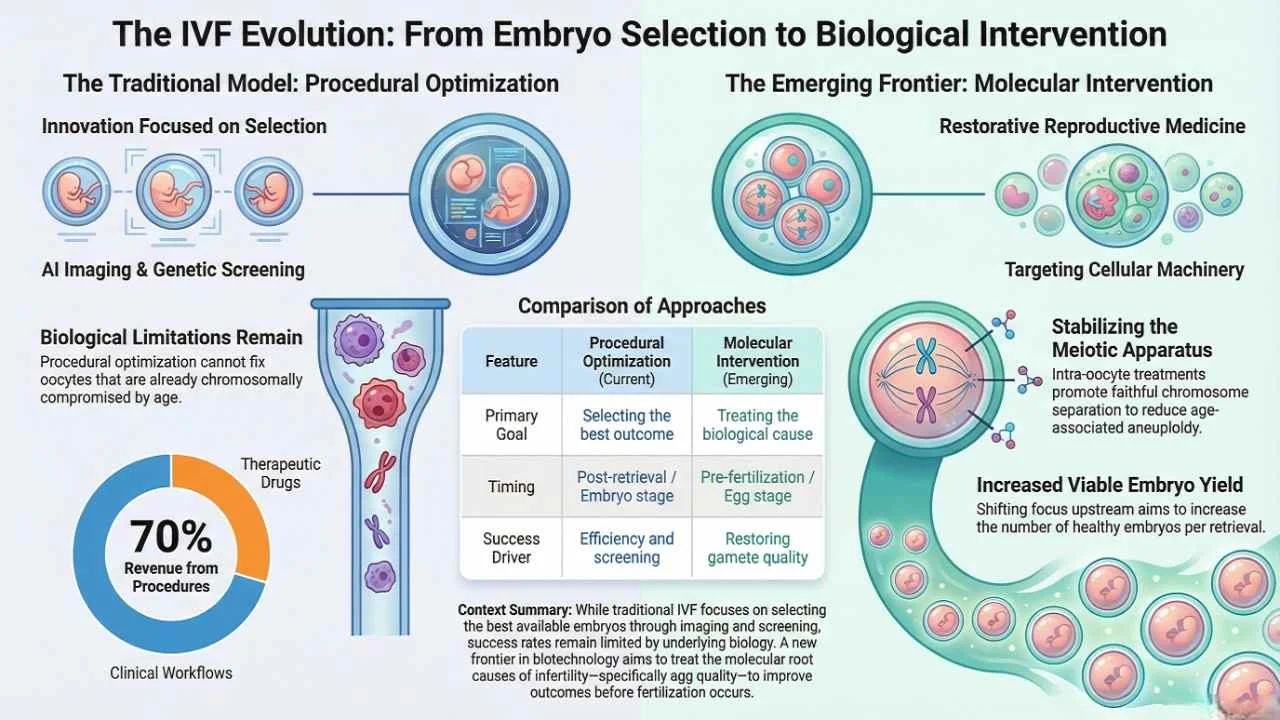
Yet, despite these advancements, In Vitro Fertilization (IVF) success rates remain stubbornly modest. The industry is now standing at a critical inflection point, moving away from the "selection-based" paradigm toward a new era of biological intervention. This shift represents the true Molecular Frontiers in IVF Success, where the focus is no longer just on choosing the best embryo, but on correcting the cellular mechanisms that cause failure in the first place.
Optimization and selection have historically characterized innovation in assisted reproductive technology (ART). Clinicians and embryologists have spent years refining the "procedural mechanics" of the laboratory.
This includes:
While these advances have undoubtedly improved laboratory efficiency and expanded access to care, they largely work around the underlying biological problem rather than addressing it. As the sources indicate, no amount of sophisticated selection can compensate for a cohort of oocytes that is already chromosomally compromised due to maternal age. This realization has prompted a strategic shift toward Molecular Frontiers in IVF Success, targeting the upstream biological processes that determine embryo viability.
The primary biological bottleneck in modern fertility care, particularly for older patients, is maternally inherited aneuploidy. This condition arises from errors in chromosome segregation during oocyte meiosis, leading to embryos that are non-viable, prone to miscarriage, or fail to implant entirely. In the traditional model, these errors are viewed as an inevitable consequence of aging, something to be screened out rather than treated.
However, a burgeoning sector of biotechnology is now investigating whether the molecular architecture of the egg itself can be stabilized. Instead of post-hoc selection, researchers are looking at the meiotic apparatus of the oocyte to prevent errors before or during maturation.
This approach aims to:
By intervening in these fundamental processes, the industry is exploring Molecular Frontiers in IVF Success that could materially change the logic of fertility treatment from one of redistribution of probability to one of actual biological improvement.
The transition from procedural optimization to biological intervention has birthed a new category: restorative reproductive medicine. This model focuses on correcting the physiological mechanisms that limit success. One of the early pioneers in this space is U-Ploid Biotechnologies, which is developing a first-in-class intra-oocyte therapeutic known as Lyvanta.
Unlike traditional pharmaceutical interventions that focus on hormonal modulation (such as gonadotropins), these new therapeutics act within the egg itself. Lyvanta, for instance, targets the cellular machinery responsible for chromosomal segregation. By promoting the faithful separation of maternal chromatids, the goal is to reduce age-associated aneuploidy prior to fertilization. This signals a broader movement within the life sciences industry to treat the root causes of infertility rather than managing its symptoms.
The current IVF model is often a grueling marathon of "repeated cycles, escalating cost, and significant emotional distress" for the patient. From a systemic perspective, this is inefficient. If therapeutic interventions can increase the number of viable embryos per retrieval, the cost-effectiveness of the entire fertility journey improves.
Furthermore, the entry of large pharmaceutical companies into this molecular space represents a significant commercial opportunity. Historically, big pharma has had a limited presence in fertility beyond basic hormonal products. The development of specialized molecular therapeutics offers a scalable path to improving outcomes that procedure-based clinics cannot achieve through manual labor or imaging alone. This evolution toward Molecular Frontiers in IVF Success is not just a scientific necessity but a commercial imperative for a sector long constrained by biology.
While the promise of molecular-level therapeutics is profound, the path to clinical adoption is fraught with hurdles. The field must navigate:
Despite these challenges, the acceleration of investment and academic collaboration suggests that we are witnessing the first credible move toward treating the root causes of infertility.
The advancement of reproductive medicine is no longer a matter of simply building better incubators or sharper microscopes. The future lies in the Molecular Frontiers in IVF Success, where biotechnology and translational science converge to restore biological function. By moving fertility care "upstream", from selecting outcomes to intervening in the processes that determine them, we offer patients more than just better odds; we offer a solution to the biological limitations of age.
