The female microbiome plays a crucial role in fertility, IVF outcomes, and pregnancy stability. Emerging research shows that gut, vaginal, and uterine microbes influence inflammation, immune tolerance, hormonal balance, and embryo implantation. Understanding this connection is transforming reproductive medicine and guiding more personalized fertility care.



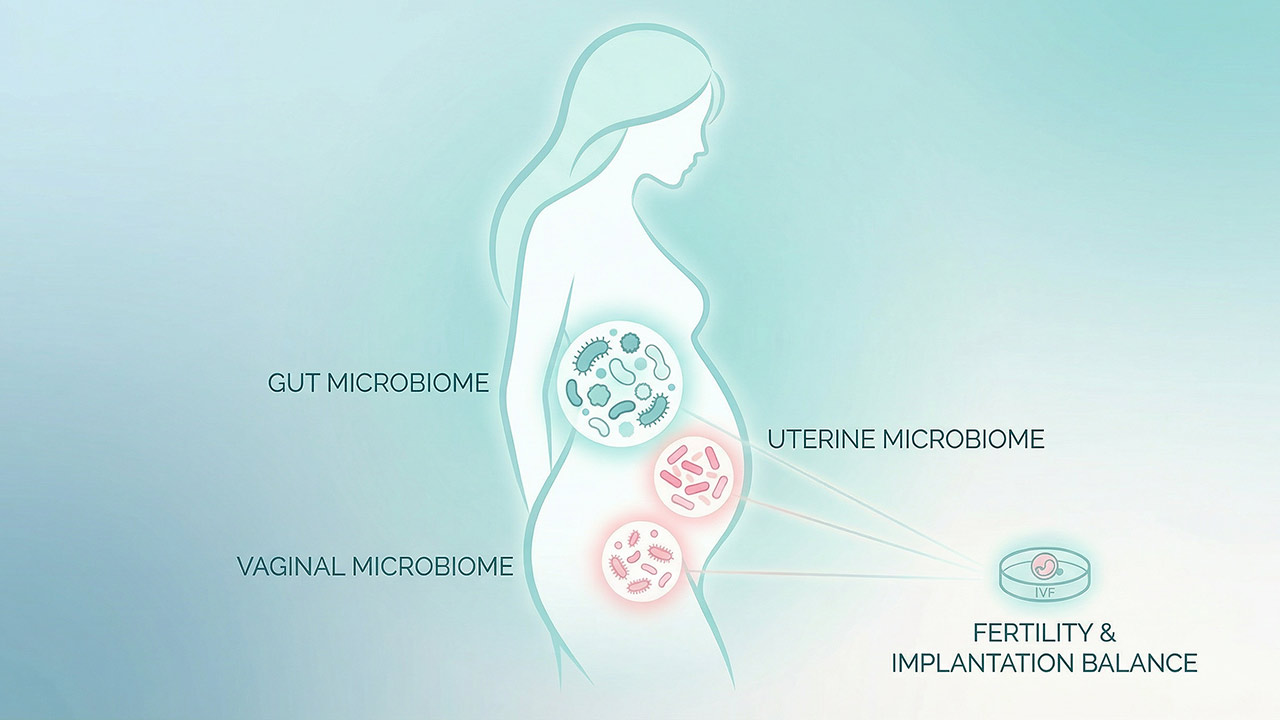
Females trying to conceive often focus on diet, hormones, and timing, yet an unexpected factor may be shaping their journey: the microbiome. The latest research indicates that the microbes living in the vaginal, gut, and uterine environments can influence everything from natural conception to embryo implantation. At its simplest, the microbiome is not a side topic anymore. It is an essential part of reproductive physiology. It influences immune response, inflammation, hormone processing, and the ability of an embryo to implant. The latest research consistently points to one conclusion: microbial stability matters long before conception begins. As more findings emerge, experts now emphasize that the female microbiome and fertility are closely interconnected.
One of the clearest patterns emerging in studies is the profound relationship between gut health and reproductive function. When the gut microbiome is balanced, it produces short-chain fatty acids that support glucose regulation, strengthen gut barrier integrity, and encourage a tolerant, pregnancy-friendly immune response.
Chronic stress, obesity, a high-fat diet, or inactivity disrupt the gut microbiome, leading to an increase in inflammation and its frequent spread. Several studies now show links between gut dysbiosis and higher risks of implantation problems, early miscarriage, gestational diabetes, and preeclampsia.
Inside the vagina, the balance is even more delicate. A healthy vaginal microbiome is typically dominated by Lactobacillus species such as Lactobacillus crispatus. These bacteria maintain a low pH and prevent harmful microbes from proliferating. When Lactobacillus levels drop, the pH rises, anaerobic bacteria grow, and inflammation follows a pattern strongly tied to bacterial vaginosis (BV) and reduced implantation rates in IVF.
Studies now show:
These aren’t small effects. In repeated implantation failure cases, vaginal microbiome testing is increasingly part of the diagnostic workflow in many advanced fertility centers.
The upper reproductive tract, cervix, endometrium, and fallopian tubes contain far fewer bacteria, making it harder to study. However, new sequencing data suggests that having more Lactobacillus in the endometrium may help embryos stick better, while higher levels of bacteria like Gardnerella or Streptococcus could cause ongoing inflammation and lower success rates in IVF.
This area remains technically challenging, but the direction of the data is consistent enough that fertility specialists have begun to treat microbial balance as part of endometrial preparation in difficult cases.
Researchers are now exploring several avenues to intentionally shift microbial balance;
Some already usable in practice, while others are still experimental.
The relationship between the microbiome and IVF extends beyond the vagina. Gut inflammation influences hormonal balance and glucose regulation. Vaginal dysbiosis affects embryo implantation. Endometrial microbial shifts may impact placental development.
Many fertility clinics are not yet routinely screening for microbiome imbalance, but the practice is beginning to change, particularly for patients with unexplained infertility or repeated IVF failures.
The female microbiome and fertility appear to be far more interconnected than once believed. It affects immune tolerance, inflammation, embryo implantation, and pregnancy stability. While much of the science is still evolving, the direction is unmistakable: microbial balance is essential to fertility, and understanding it can help clinicians personalize care for better outcomes.
As research expands, microbiome-guided fertility treatment may become one of the most transformative frontiers in reproductive medicine, offering new hope for individuals and couples navigating the journey to parenthood.
Disclaimer: This article is for informational purposes only and does not constitute medical advice, diagnosis, or treatment. Fertility conditions vary widely, and the technologies and procedures described may not be appropriate for every individual. Always consult a qualified healthcare professional, such as a gynecologist or reproductive endocrinologist, for personalized medical guidance. Never disregard medical advice or delay seeking care because of information presented here.
